Cardiac Sarcoidosis: Role of Multimodality Imaging for Diagnosis and Treatment
Deepti Bhandare1*, Anupama Kottam2
1Department of Cardiology, AdventHealth Sebring, Florida
2Department of Cardiology, Detroit Medical Center, Michigan
Abstract
The clinical presentation of cardiac sarcoidosis (CS) ranges from an incidentally discovered condition to heart failure and sudden death. The diagnosis of CS is tough, and as a result, CS is often under-recognized in clinical practice. CS is mostly noted in the setting of systemic sarcoidosis, though isolated CS can occur. Frequently clinical criteria require the diagnosis of extracardiac disease in order to establish the diagnosis of CS in the absence of having a positive endomyocardial biopsy. While endomyocardial biopsy provides a high specificity for diagnosing CS, this invasive test has a limited sensitivity. There is incomplete knowledge of disease development and a deficient consensus on the ideal methods for disease recognition. We discuss CS in general, the clinical disease, diagnostic algorithms, latest guidelines and management.
Abbreviations
CS: Cardiac Sarcoidosis; CMR: Cardiac MRI; PET: Positron Emission Tomography; EMB: Endomyocardial Biopsy; HRS: Heart Rhythm Society; ACC: American College of Cardiology; AHA: American Heart Association; LGE: Late Gadolinium Enhancement.
Introduction
Sarcoidosis is a disease process with an unidentified cause, which manifests pathologically as noncaseating granulomas with multiorgan involvement. The characteristic pattern is bilateral hilar lymph node involvement, reticular opacities in the lung and joint, eye, peripheral nervous system involvement and/or skin lesions. The incidence and prevalence (estimated at 10 to 20 per 100,000 population) of sarcoidosis are undetermined. The disease has no geographical predominance. It usually involves adolescents and young adults1.
The prevalence of Cardiac Sarcoidosis among patients with systemic sarcoidosis is reported to be 20 to 27 percent in the United States and as high as 58 percent in Japan2. It has been estimated that the lifetime risk of sarcoidosis in blacks in the United States is 2.4 percent, compared with a lifetime risk of 0.85 percent in whites27,28.
Symptoms
Symptoms of CS include palpitations, presyncope, syncope, fatigue, dyspnea, orthopnea, and sudden cardiac death. Palpitations may be caused by either supraventricular or ventricular arrhythmias. Presyncope or syncope can be caused by AV block, ventricular tachycardia, or supraventricular tachycardia. Fatigue, dyspnea, and orthopnea can reflect heart failure caused by CS34.
First degree AV block is the most common clinical presentation in patients with clinically evident CS. First-degree AV block due to disease of the AV node or bundle of His and intraventricular conduction defect is common and may progress35.
Ventricular arrhythmias (sustained or nonsustained ventricular tachycardia and ventricular premature beats [VPBs]) are the second most common clinical presentation of CS, occurring in approximately 30 percent of cases3. Supraventricular arrhythmias seen with CS include paroxysmal atrial tachycardia, atrial flutter, atrial fibrillation, and sinus arrest secondary to granulomatous involvement of the sinus node34.
Sudden death due to ventricular tachyarrhythmia or conduction block accounts for 25 to 65 percent of deaths caused by CS. Implantation of ICD should be considered in patients with patients at risk for ventricular arrhythmias and sudden cardiac death. A recent study reported reduced mortality rates in patients receiving appropriate immunosuppressant therapy for cardiac sarcoidosis versus those without treatment20.
Age and lack of pacemaker or defibrillator were the significant predictors of mortality for CS. Decreased LVEF of less than 40% was associated with worse prognosis30. Reduced lung diffusion capacity (<35% of predicted) and 6-minute walk distance of less than 300 meters were associated with reduced survival in the overall precapillary cohort. Preserved FEV1/FVC ratio was also identified as an independent risk factor for worsened outcomes32.
CS can cause either a dilated cardiomyopathy or a restrictive cardiomyopathy which can lead to HF with preserved ejection fraction. Few patients with CS present right-sided heart failure, due to sarcoid-related inflammation or scar affecting the right ventricle36.
Diagnostic Tests
Echocardiographic findings in patients with CS are varied. Focal areas of edema can result in increased wall thickness mimicking hypertrophic cardiomyopathy and focal areas of akinesis, dyskinesis, wall thinning [Figure 1]. Among patients with CS, LVEF can be either preserved or reduced and there is evidence of diastolic dysfunction.
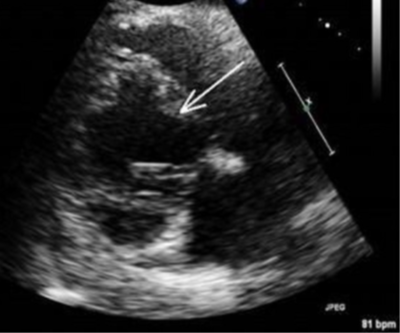
Figure 1: Transthoracic echocardiogram in parasternal long-axis view demonstrating septal thinning and akinesis (white arrow) in systolic frame. [Adapted from Bhandare D et al14].
While echocardiography has a limited sensitivity for detecting CS compared with cardiac magnetic resonance [CMR], it can be useful for evaluating the effects of pulmonary sarcoidosis and CS on hemodynamics and cardiac structure and function including left and right ventricle size and function, valve function, and estimating right heart and pulmonary pressures.
The reduced global longitudinal strain may be present in CS despite preserved ejection fraction, and reduction in longitudinal strain magnitude may vary inversely with late gadolinium enhancement (LGE) burden [Figure 2]3,14.
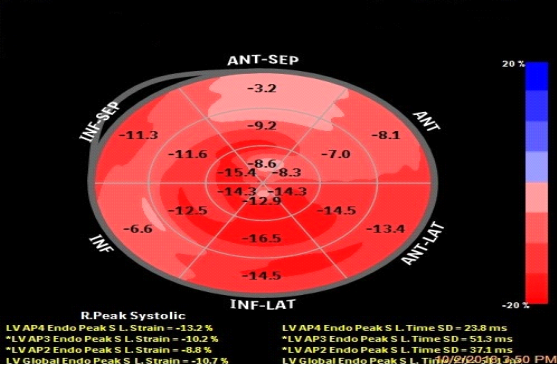
Figure 2: Echocardiographic strain imaging showing decreased longitudinal strain pattern in the anteroseptal region (Philips EPIQ). (Anteroseptal, anterior, anterolateral, inferolateral, inferior, inferoseptal wall). [Adapted from Bhandare et al14]
Echocardiography has low sensitivity of 25 to 65 percent for detection of CS as compared to CMR or 18F-fluorodeoxyglucose Positron Emission Tomography (FDG PET)21. Despite a high positive predictive value (84 percent), echocardiography had a low sensitivity (27 percent) to detect CS and, when added to the initial screening based on cardiac history and ECG, did not provide any improvement in sensitivity. Based on the above results, in patients with extracardiac sarcoidosis who have symptoms or signs of possible cardiac involvement, echocardiography should not be used as a screening test, as a negative echocardiogram cannot be used to rule out cardiac involvement22.
The CMR method for detecting CS is the identification of late gadolinium enhancement [LGE], which is most commonly multifocal and involves the midventricular wall or subepicardium [Figure3]. Of note, no specific pattern of LGE is pathognomonic for CS, therefore, careful interpretation in the context of other clinical features is required.
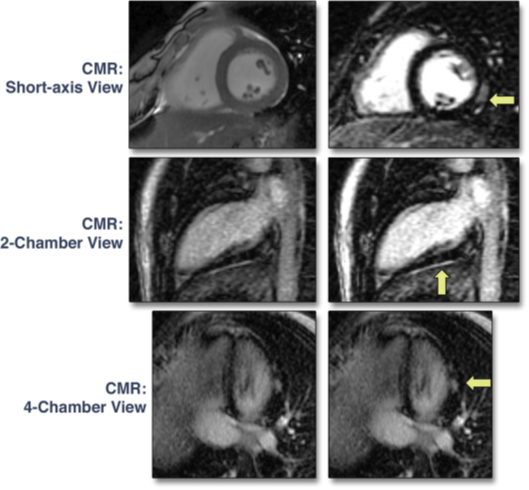
Figure 3: Cardiac magnetic resonance (CMR) in a patient with sarcoidosis showing subepicardial late gadolinium enhancement in the left ventricular lateral wall in 2-chamber, 4-chamber, and short-axis views and matching steady-state free precession cine imaging (arrows) [ Adapted from Kournaos V et al3]
The following are typical although not specific LGE patterns in patients with CS:
• Multifocal areas of LGE4
• Sub-epicardial and mid-myocardial LGE, although some patients may have sub-endocardial involvement in a pattern like myocardial infarction23
• Areas of increased T2 signal often representing inflammation23
• Direct LGE extension from the left ventricle, across the interventricular septum, into the right ventricle24
The main strength of CMR is the high negative predictive value for excluding CS when no LGE is detected5. Moreover, contrast MRI is useful for outcome prediction and risk stratification regarding need for ICD-implantation. Presence and extent of ventricular Gadolinium enhancement is associated with increased risk for ventricular arrhythmias/ventricular fibrillation and sudden cardiac death15. This risk is even further increased if right ventricular Gadolninium enhancement is present in MRI16.
Ischemic cardiomyopathy, which is a common differential diagnosis, is characterized by subendocardial and/or transmural LGE. In comparison, isolated mid-wall or epicardial enhancement is strongly suggestive of sarcoid cardiomyopathy. The territorial vascular distribution of ischemic is quite typical versus the varied distribution pattern in cardiac sarcoidosis.
18F-fluorodeoxyglucose positron emission tomography (FDG-PET) can detect active myocardial inflammation, which, in the appropriate clinical context, can be used to determine the likelihood of CS37 [Figure 4]. FDG uptake by the heart is non-specific for CS and can be seen in other inflammatory myocardial diseases and hibernating myocardium. Extra cardiac FDG-PET images are strongly recommended when there are no prior data regarding the presence or disease activity of extra cardiac sarcoidosis6.
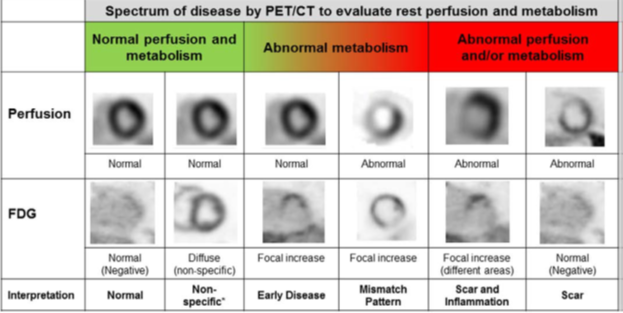
Figure 4: Spectrum of cardiac sarcoidosis by cardiac positron emission tomography (PET). *Likely failure to suppress 18F-fluorodeoxyglucose (FDG) uptake by normal myocardium although rarely can also be seen with diffuse disease. CT indicates computed tomography [Adapted from Blankstein R et al33].
CMR and FDG-PET are the two imaging modalities that appear to have the highest sensitivity for detection of CS. CMR is more likely to provide information regarding the occurrence and degree of edema and scar and PET offers information regarding the presence, extent, and severity of myocardial inflammation7.
An endomyocardial biopsy [EMB] is recommended when histologic confirmation of non-caseating granulomas from extra cardiac source is lacking [Figure 5] EMB has a low sensitivity of less than 30 percent and its use is limited by false negative results owing to sampling error that may occur due to the patchy distribution of disease8.
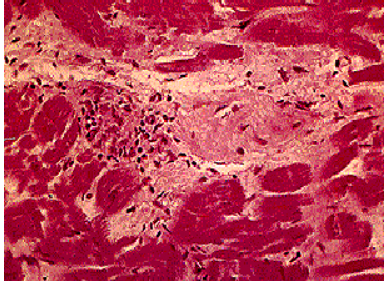
Figure 5: Non caseating granulomatous inflammation within the myocardial wall [Adapted from UpToDate].
Diagnosis of Cardiac Sarcoidosis
A definite diagnosis of CS is established by detection of noncaseating granuloma on histologic examination of myocardial tissue with no alternative cause identified. The term probable CS denotes a likelihood of CS ≥50 percent. This term recognizes the inherent uncertainty that is often clinically present when evaluating patients with suspected CS5.
Since the histologic findings are not pathognomonic, some experts consider the diagnosis of CS "highly probable" if myocardial noncaseating granulomas are detected22,30.
The diagnosis of CS is frequently uncertain. Categories (highly probable, probable, and possible CS) may be used in cases in which there is uncertainty regarding the diagnosis of CS. This classification system, while not widely used for evaluating the likelihood of CS, has been developed based on the World Association of Sarcoidosis and Other Granulomatous Diseases (WASOG) organ assessment instrument, which is used to determine the probability of sarcoidosis organ involvement29.
For patients with uncertain diagnosis of CS, the presence of one or more of these clinical findings suggests a higher likelihood of CS5.
â Unexplained reduced LVEF (<40 percent)
â Unexplained sustained VT (spontaneous or induced)
â AV block: Mobitz type II second degree or third degree
Comparison of Major Society Guidelines
The most commonly used clinical criteria for diagnosing CS are the revised Japanese Ministry of Health and Welfare (JMHW) criteria9,10,20 and the HRS Expert Consensus Statement5. Both guidelines provide a histologic pathway whereby a definitive diagnosis of CS can be established by an EMB, which reveals noncaseating granulomas. In patients who do not have a positive EMB, these criteria require a diagnosis of extracardiac sarcoidosis (for the JMHW, this can be either clinical or histologic, while for the HRS criteria, histologic diagnosis of extracardiac sarcoidosis is required) in conjunction with other criteria.
This led to a diagnostic conundrum for isolated CS due to the low sensitivity and associated risks of EMB regardless of developments in image mediated protocols. The Japanese Ministry of Health and Welfare guidelines for sarcoidosis in 2015 proposed a novice guideline in 2015 that replaces histologic confirmation with PET and cardiac MRI as a major criterion11. The criteria have been classified as Major and Minor and require the presence of two of the five major or one major and 2 minor for the diagnosis of CS.
The major criteria are:
a. Late myocardial enhancement on contrast MRI.
b. Abnormal cardiac uptake in FDG-PET
c. Impaired ventricular function with regional wall motion abnormality.
d. Basal thinning of the septal wall or morphologic ventricular abnormality (ventricular aneurysm, wall thinning)
e. Advanced AV block or sustained ventricular tachycardia
The minor criteria were noted to be the following.
a. Nonsustained ventricular tachycardia, multifocal PVCS, bundle branch block, axis deviation.
b. Defect on myocardial perfusion scintigraphy
c. Endomyocardial biopsy: interstitial fibrosis or monocyte infiltration over moderate grade.
Differential Diagnosis
An essential part of the diagnosis of sarcoidosis is the exclusion of alternative possibilities, and several settings are particularly prone to diagnostic difficulty. Mycobacterial infection, fungal infections(histoplasmosis, blastomycosis, Pneumocystis jirovecii), HIV infection, Loffler’s syndrome, hypersensitivity pneumonitis, pneumoconiosis, drug-induced hypersensitivity, pulmonary histiocytic disorders, foreign body granulomatosis, diseases associated with vascular inflammation( granulomatosis with polyangiitis (Wegener’s), eosinophilic granulomatosis with polyangiitis (Churg-Strauss), and pulmonary lymphomatoid granulomatosis25,26.
CS needs to be distinguished from myocarditis, Arrhythmogenic right ventricular cardiomyopathy (ARVC). ARVC is often isolated to the right ventricle and spares the septum, while patients with CS and right heart myocardial involvement often also have significant septal and/or left heart infiltration. While some patients can have left-sided arrhythmogenic cardiomyopathy, the pattern of late gadolinium enhancement (LGE) on MRI between this condition and CS is different (left-sided arrhythmogenic cardiomyopathy often has a large amount of circumferential and often contiguous subepicardial LGE)31.
Management
Glucocorticoid therapy has been the most commonly used immunosuppressive agent. The optimal dose of glucocorticoid therapy for CS is not known, and choosing a dose requires balancing the risk of side effects with the likelihood of response12. In patients with significant inflammation, FDG-PET imaging at approximately six months of therapy may be reasonable to assess treatment response.
Patients are typically started on 60 mg/day of prednisone and gradually lowered to a maintenance dose of 10 to 15 mg/day over one year. Patients who have resolution of inflammation can develop recurrence. Hence, a low dose of glucocorticoid therapy like Prednisone 5 mg for at least one year may be recommended, especially if low-dose glucocorticoid therapy is well tolerated. Thus, glucocorticoid treatment should be continued for at least one to two years. In patients with significant inflammation, FDG-PET imaging at approximately six months of therapy may be reasonable to assess treatment response. If serial evaluations demonstrate that the disease is dormant, glucocorticoids may be tapered and eventually discontinued. Strict vigilance must be kept or the rest of the patient's life, as relapses are common after tapering of glucocorticoid therapy. Any evidence of recurrence may be handled by reinstituting or increasing prednisone to 40 to 60 mg/day.
The common side effects of prolonged steroid use are hypertension, diabetes, lower resistance to infection, myopathy, depression and osteoporosis.
Steroid-sparing agents such as methotrexate, azathioprine, infliximab, or mycophenolate mofetil have been used as add-on therapy or as an alternative owing to the side effect profile of glucocorticoids. These agents may worsen heart failure, and thus should be used with great caution in patients who have volume overload. Appropriate immuno-suppressant therapy for cardiac sarcoidosis is associated with slowed disease progression and reduced long-term mortality rates20,21. Establishing an appropriate immunosuppressant drug therapy in individual patients should be performed under repeated PET control studies, aiming the disappearance of inflammatory cardiac sites with increased FDG-uptake.
A permanent pacemaker is indicated in patients with complete AV block, or other high-grade conduction system disease, even if transient. In such cases, implantation of an ICD, rather than a pacemaker alone can be considered13. Several studies revealed the independent predictors of life-threatening ventricular arrhythmias and sudden cardiac death as the following: extent of myocardial late gadolinium enhancement15,21 previous/presentation with ventricular tachycardia, and impaired left ventricular ejection fraction17. This risk for sudden cardiac death is even further increased if right ventricular Gadolinium enhancement is present in MRI16.
The 2014 HRS Expert Consensus Statement on the Diagnosis and Management of Arrythmias associated with CS5 recommended that for patients with one or both (i) LVEF 36% to 49% and (ii) LVEF < 40% ICD implantation is recommended [class II B recommended]. In patients with normal LVEF consider CMR and if LGE is noted then do electrophysiological study [EPS]. If EPS is positive, then consider ICD implantation (class IIa recommendation). ICD implantation is not recommended in patients with normal LVEF/RVEF and a negative electrophysiology study (regardless if LGE is noted on CMR).
The 2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death recommend ICD implantation (class IIa recommendation) in patients with evidence of extensive myocardial scar by cardiac MRI or PET scan with LVEF greater than 35%18,19.
References
- Thomas KW, Hunninghake GW. Sarcoidosis. JAMA. 2003; 289: 3300.
- Okada DR, Bravo PE, Vita T, et al. Isolated cardiac sarcoidosis: A focused review of an under-recognized entity. J Nucl Cardiol. 2018; 25: 1136.
- Kouranos V, Tzelepis GE, Rapti A, et al. Complementary Role of CMR to Conventional Screening in the Diagnosis and Prognosis of Cardiac Sarcoidosis. JACC Cardiovasc Imaging. 2017; 10: 1437.
- Greulich S, Deluigi CC, Gloekler S, et al. CMR imaging predicts death and other adverse events in suspected cardiac sarcoidosis. JACC Cardiovasc Imaging. 2013; 6: 501.
- Birnie DH ,William S, Frank B, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014; 11: 1305.
- Chareonthaitawee P, Beanlands P, Chen W, et al. Joint SNMMI-ASNC Expert Consensus Document on the Role of 18F-FDG PET/CT in Cardiac Sarcoid Detection and Therapy Monitoring. J Nucl Med. 2017; 58: 1341.
- Vita T, Okada DR, Veillet-Chowdhury M, et al. Complementary Value of Cardiac MRI and PET/CT in the Assessment of Cardiac Sarcoidosis. Circ Cardiovasc Imaging. 2018; 11: e007030.
- Uemura A, Morimoto S, Hiramitsu S, et al. Histologic diagnostic rate of cardiac sarcoidosis: evaluation of endomyocardial biopsies. Am Heart J. 1999; 138: 299.
- Japan Society of Sarcoidosis and Other Granulomatous Disorders. Diagnostic standard and guidelines for sarcoidosis. Jpn J Sarcoidosis Granulomatous Disord. 2007;27: 89-102.
- Au K, Wong D, Newbigin K, at al. Multimodality Imaging to Diagnose Isolated Cardiac Sarcoidosis and Determine Regional Inflammatory Activity Levels. CASE. 2017 Sep 1; 1(5): 171-175.
- Sano M, Satoh H, Suwa K, et al. Intra-cardiac distribution of late gadolinium enhancement in cardiac sarcoidosis and dilated cardiomyopathy. World J Cardiol. 2016; 8: 496-503.
- Hamzeh NY, Wamboldt FS, Weinberger HD, et al. Management of cardiac sarcoidosis in the United States: a Delphi study. Chest. 2012; 141: 154.
- ACC/AHA/HRS 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: a report of the ACC/AHA Task Force on Practice Guidelines. J Am Coll Cardiol. 2008.
- Bhandare D, Kottam A. Atypical presentation of cardiac sarcoidosis: role of multimodality imaging and review of literature. BMJ Case Rep. 2020 Feb 10; 13(2). pii: e232047. doi: 10.1136/bcr-2019-232047.
- Ekström K, Nordenswan HK. J Am Heart Assoc. 2016; 5: e003040 and Circ ArrhythmElectrophysiol.2018; 11: e006145.
- Crawford T. https://doi.org/10.1161/CIRCEP.113.000156Circulation: Arrhythmia and Electrophysiology. 2014; 7: 1109–1115.
- Nordenswan HK. Circ ArrhythmElectrophysiol. 2018; 11: e006145.
- Al-Khatib SM. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. J Am Coll Cardiol. 2017; 25: 318–320.
- Birnie D, Ha A, Kron J. Which patients with cardiac sarcoidosis should receive implantable Cardioverter-Defibrillators: Some Answers but Many Questions Remain. Circ Arrhythm Electrophysiol. 2018 Aug; 11(8): e006685. doi: 10.1161
- Soejima K, Yada H. The work-up and management of patients with apparent or subclinical cardiac sarcoidosis: with emphasis on the associated heart rhythm abnormalities. J Cardiovasc Electrophysiol. 2009; 20: 578.
- Ekstrom K, Lehtonen J, Nordenswan HK, et al. Sudden death in cardiac sarcoidosis: an analysis of nationwide clinical and cause-of-death registries. European Heart Journal. 2019; 40: 3121–3128. doi:10.1093.
- Freeman AM, Curran-Everett D, Weinberger HD, et al. Predictors of cardiac sarcoidosis using commonly available cardiac studies. Am J Cardiol. 2013 Jul 15; 112(2): 280-5. doi: 10.1016
- Mehta D, Lubitz SA, Frankel Z, et al. Cardiac involvement in patients with sarcoidosis: diagnostic and prognostic value of outpatient testing. Chest. 2008 Jun; 133(6): 1426-1435. doi: 10.1378
- Patel MR, Cawley PJ, Heitner JF, et al. Detection of myocardial damage in patients with sarcoidosis. Circulation. 2009 Nov 17; 120(20): 1969-77. doi: 10.1161
- Sharma S. Cardiac imaging in myocardial sarcoidosis and other cardiomyopathies. Curr Opin Pulm Med. 2009; 15: 507.
- Gulel O, Camlidag I, Elmali M. Successful Medical Management of a Rare Loeffler Endocarditis Case. Korean Circ J. 2018 Jul; 48(7): 655-657. doi: 10.4070.
- Haramati LB, Lee G, Singh A, et al. Newly diagnosed pulmonary sarcoidosis in HIV-infected patients. Radiology. 2001; 218: 242.
- Rybicki BA, Major M, Popovich J Jr, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997; 145: 234.
- Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med. 1999; 160: 736.
- Zhou Y, Lower EE, Li HP, et al. Cardiac Sarcoidosis: The Impact of Age and Implanted Devices on Survival. Chest. 2017; 151: 139.
- Philips B, Madhavan S, James CA, et al. Arrhythmogenic right ventricular dysplasia/cardiomyopathy and cardiac sarcoidosis: distinguishing features when the diagnosis is unclear. Circ Arrhythm Electrophysiol. 2014; 7: 230.
- Shlobin OA, Kouranos V, Barnett SD, et al. Physiological Predictors of Survival in Patients with Sarcoidosis Associated Pulmonary Hypertension: Results from an International Registry. Eur Respir J. 2020 Mar 26. pii: 1901747. doi: 10.1183/13993003
- Ron Blankstein MD, Alfonso H, Waller MD. Evaluation of Known or Suspected Cardiac Sarcoidosis. Circ Cardiovasc Imaging. 2016; 9: e000867. DOI: 10.1161
- Hulten E, Aslam S, Osborne M, et al. Cardiac sarcoidosis-state of the art review. Cardiovasc Diagn Ther 2016; 6: 50.
- Nery PB, Beanlands RS, Nair GM, et al. Atrioventricular block as the initial manifestation of cardiac sarcoidosis in middle-aged adults. J Cardiovasc Electrophysiol. 2014; 25: 875.
- Lønborg J, Ward M, Gill A, et al. Utility of cardiac magnetic resonance in assessing right-sided heart failure in sarcoidosis. BMC Med Imaging. 2013; 13: 2.
- Youssef G, Leung E, Mylonas I, et al. The use of 18F-FDG PET in the diagnosis of cardiac sarcoidosis: a systematic review and metaanalysis including the Ontario experience. J Nucl Med. 2012; 53: 241.
