Dilemma During the Pandemic- Hickam’s Dictum or Occam’s Razor?
Akanksha Agrawal1*, Deepanshu Jain2, Jefferson Baer1, Michael McDaniel1
1Division of Cardiology, Emory University School of Medicine, Atlanta, GA, USA
2Center for Interventional Endoscopy, AdventHealth, Orlando, FL, USA
Abstract
Coronavirus Disease 2019 (COVID-19) is a primary respiratory illness with various cardiac manifestations. This case describes a patient presenting as acute myocardial infarction (AMI) with cardiogenic shock and acute hypoxic respiratory failure secondary to COVID-19 complicated by acute embolic stroke.
Introduction
A 14th-century philosopher, William of Ockham once stated “Pluralitas non est ponenda sine necessitate” translating to the well-known Ockham’s razor “entities should not be multiplied unnecessarily”. A counterargument to this in the medical field is the Hickam’s dictum stating “A man can have as many diseases as he damn well pleases”. As such, we present a patient presenting as acute myocardial infarction with cardiogenic shock, undergoing revascularization and developing acute hypoxic respiratory failure secondary to COVID-19 resulting in multiorgan failure. This case highlights the challenges in diagnosis and management and the risks associated with delays in care.
Case
A 67 year old man with past medical history of hypertension presented to an outside hospital emergency department without primary percutaneous coronary intervention (PCI) capability with chest pain and difficulty breathing. He had ongoing constant chest pain for almost 23 hours prior to presentation, and fever, chills, cough, and shortness of breath for few days prior to presentation. His initial blood pressure was normal, and the electrocardiogram was concerning for recent anterior ST-segment elevation myocardial infarction (STEMI) with q waves in V2-V4 (Figure 1) and a peak troponin of 125 ng/ml. A chest x-ray demonstrated bilateral patchy infiltrates concern for pneumonia or pulmonary edema. He was given aspirin 325 mg, heparin 5000 units, and transferred to our center for a left heart catheterization (LHC) and primary PCI.
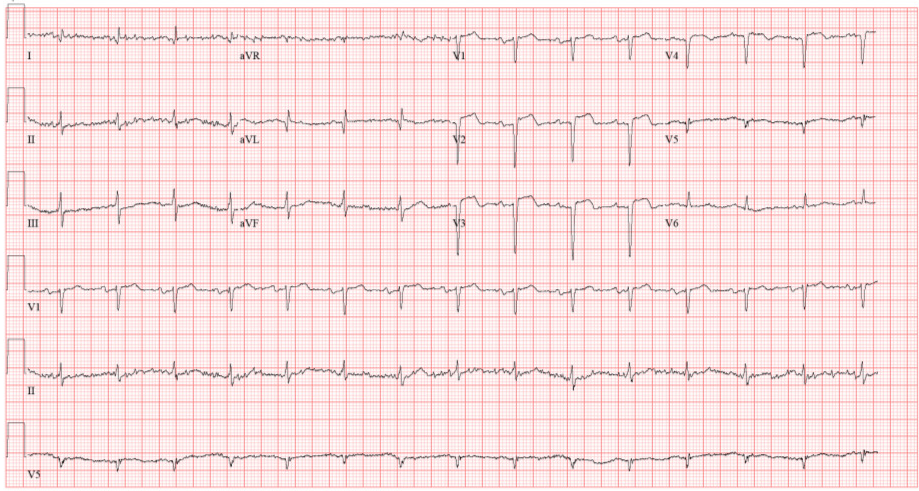
Figure 1: Electrocardiogram from the first presentation showing ST elevation in lead V2-V4 with the presence of q wave in V2-V4
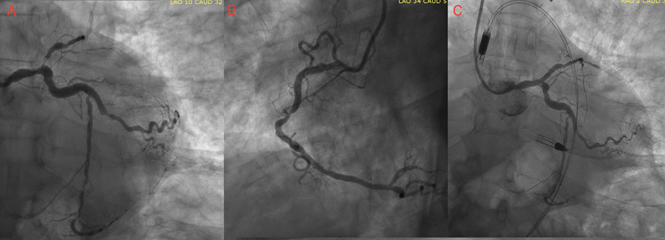
Figure 2: A. Left anterior oblique view of the coronary angiogram depicting disease in the left main, left anterior descending and left circumflex vessels. B. Left anterior oblique view of the coronary angiogram depicting disease in the right coronary artery. C. Coronary angiogram after revascularization with drug eluting stent in LM-LAD and Impella in place.
On reaching our center an hour later, his temperature was 38.7 degrees Celsius and he was breathing at 30/min with saturation 100% on a non-rebreather mask. Emergent coronary angiography demonstrated a 70% distal left main disease and a 99% ostial left anterior descending (LAD) culprit lesion with occlusion in the mid-LAD and thrombolysis in myocardial infarction- 1 (TIMI-1) flow. The left circumflex had 70 % diffuse disease and the right coronary artery (RCA) was the dominant vessel with 70-80% mid lesion. (Figure 2) The left ventricular end diastolic pressure (LVEDP) was severely elevated at 33mmHg and left ventriculography showed a severely reduced left ventricular ejection fraction (LVEF) of around 20% with severe anterior and apical hypokinesis. During angiography, his blood pressure (BP) dropped to 86/55 mmHg. Given the anterior STEMI, multivessel coronary artery disease (CAD), severely reduced LVEF, elevated LVEDP, and shock an Impella CP device was inserted via right femoral access. A 3.25x28 mm Xience drug eluting stent (DES) was deployed to the ostial left main (LM) and proximal LAD and the ostial LM post dilated to 4.5X15 mm. (Figure 2C) Right heart catheterization at the conclusion of the case showed right atrial (RA) pressure of 15 mmHg, pulmonary artery (PA) 38/21 (mean 27 mmHg), pulmonary capillary wedge pressure (PCWP) of 26 mmHg, cardiac index 1.3 and cardiac output of 2.7 L/min. Given the persistent cardiogenic shock, extracorporeal membrane oxygenation (ECMO) was considered but would have required transport to yet another facility. The patient was admitted to the cardiac care intensive unit as a person under investigation (PUI) for COVID-19 given his fever, cough, and shortness of breath and a nasopharyngeal swab to rule out COVID-19 was performed. The post-procedure chest x-ray (CXR) showed bilateral patchy opacities (Figure 3). Laboratory investigations illustrated in Table 1.
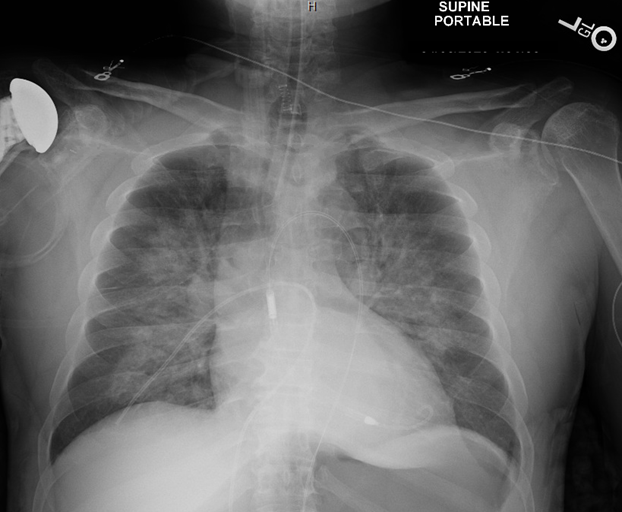
Figure 3: Chest radiograph showing bilateral patchy opacities concerning for mutifocal pneumonia or pulmonary edema
Table 1. Initial laboratory work up with results
|
Laboratory Test (Unit) |
Value (normal range) |
Laboratory Test (Unit) |
Value (normal range) |
|
Hemoglobin (g/dL) |
13 (12.9- 16.1) |
Fibrinogen Activity level (mg/dL) |
571 (200-393) |
|
White Blood Count (103/mcL) |
16.8 (4.2-9.1) |
Alanine Aminotransferase (unit/L) |
101 (7-52) |
|
Platelet count (103/mcL) |
311 (150-400) |
Aspartate Aminotransferase (unit/L) |
764 (13-39) |
|
Sodium (mmol/L) |
137 (136-145) |
Creatine Kinase (unit/L) |
4977 (30-223) |
|
Potassium (mmol/L) |
4.7 (3.5- 5.1) |
Brain Natriuretic Peptide (pg/mL) |
810 (<=99) |
|
Blood Urea Nitrogen (mg/dL) |
22 (7-25) |
Troponin (ng/mL) |
>70 (<=0.04) |
|
Creatinine (mg/dL) |
1.53 ( 0.7- 1.3) |
Triglyceride (mg/dL) |
301 (<= 149) |
|
D-dimer (ng/mL) |
4724 (<= 574) |
HDL Cholesterol (mg/dL) |
<3 (23-92) |
|
Lactate Dehydrogenase (units/L) |
1522 (140-271) |
C-reactive protein (mg/L) |
69.4 (<=10) |
|
Hemoglobin A1c (%) |
6.0 (<=5.6) |
Interleukin 6 (pg/mL) |
135 (<=5) |
|
Prothrombin Time (seconds) |
15.2 (9.4- 12.5) |
Activated Prothrombin time (APTT) (seconds) |
44.4 (25.1- 36.5) |
|
International Normalized Ratio (INR) |
1.31 |
|
|
Within a few hours, the patient had progressively increasing work of breathing and the degree of hypoxia seemed out of proportion to his LVEDP and overall volume status. His arterial blood gas while on non-rebreather mask showed pH of 7.43, pCO2 28 mmHg and pO2 of 50 mmHg and he was intubated, requiring 100% FiO2 and 10 of positive end expiratory pressure (PEEP) on mechanical ventilation. The following day, he required increasing vasopressor and inotropic requirements for worsening shock of mixed cardiogenic and distributive shock physiology and paralysis for refractory hypoxia. His COVID-19 polymerase chain reaction (PCR) study resulted positive. He was considered for remdesivir but was not able to receive due to worsening renal function. He ultimately required continuous renal replacement therapy (CRRT) for worsening kidney failure. His clinical course was further complicated by multiple acute embolic strokes, and he finally succumbed to multi organ failure on Day 11 of hospitalization.
Discussion
Our case highlights the challenges in the management of acute myocardial infarction complicated by severe acute respiratory syndrome (SARS-CoV-2) from coronavirus (COVID-19). In addition, this case highlights many of the potential cardiovascular complications from COVID-19 infection. Moreover, this case serves as a warning for the worse outcomes that can result from delayed care in myocardial infarction during this pandemic.
Accurate diagnosis and timely care can be challenging for patients with acute myocardial infarction and COVID-19 infection. In this case, the patient presented with SOB and cough with bilateral interstitial infiltrates noted on CXR. As the respiratory status worsened, it was challenging to determine the precise reason for clinical deterioration despite invasive hemodynamic monitoring. The high LVEDP and low cardiac output (CO) suggested pulmonary edema from acute decompensated heart failure (ADHF), but the hypoxia seemed out of proportion to the LVEDP and failed to respond to Impella support, revascularization, and diuresis. With time, it became clear the major etiology for the hypoxia was severe acute respiratory syndrome from COVID-19. In addition, the shock that developed also had a mixed etiology; low cardiac output from acute heart failure from acute myocardial infarction and distributive etiology from acute inflammation. Early in the hospitalization the cardiogenic etiology predominated, but as the hospitalization progressed the distributive etiology became more evident. Heightened systemic inflammation is characteristic of severe COVID-19 infection and likely resulted in low systemic vascular resistance (SVR). Various inflammatory markers such as interleukin (IL) 6, IL- 2, IL-7, tumor necrosis factor (TNF)-α, granulocyte-colony stimulating factor (G-CSF), C-reactive protein (CRP), procalcitonin, and ferritin have been found to be elevated, resulting in a cytokine storm1.
This case highlights some of the potential cardiovascular complications of COVID-19 infection. In a study including 416 hospitalized patients with COVID-19, almost 20% had cardiac injury2. Multiple studies have shown cardiac injury to be associated with higher risk of in-hospital mortality. The acute inflammation from respiratory infection may be one of the precipitating factors for acute coronary syndrome. Prior studies have demonstrated increased risk ratio [6.05 (95% confidence interval, 3.86 to 9.500] for acute myocardial infarction during first 7 days of influenza infection compared to the control interval of1 year before and after the virus test3. An acute infectious process may cause acute coronary syndrome (ACS) through acute inflammation, biomechanical stress and vasoconstriction in a milieu of chronic atherosclerotic vascular disease4. In addition, COVID-19 infection has been associated with a prothrombotic state. Whole blood thromboelastographic profiles of COVID-19 patients demonstrated hypercoagulability and has been linked to both markedly increased levels of fibrinogen and an excessive fibrin polymerization due to the infection5. There are reports of both venous thromboembolic events and acute myocardial infarction in patients with COVID-196,7. In a study of 184 ICU patients with COVID-19 pneumonia, 31% incidence of thrombotic complications including acute pulmonary embolism, deep venous thrombosis, and ischemic stroke was observed6. This prothrombotic state may have contributed to the myocardial infarction as well as the embolic strokes that complicated the hospital course. The patient had an Impella CP device, severe apical hypokinesis from anterior myocardial infarction, low ejection fraction, aortic arch atheroma, and acute coronary thrombosis, all of which may have contributed to the stroke on their own. However, a higher incidence of embolic strokes has been noted in patients with COVID-19 suggesting the prothrombotic state from the infection may have also contributed to the complication. Retrospective single-centric data of 221 patients with COVID-19 showed a 5% incidence of acute ischemic stroke, suggesting acute cerebrovascular disease is not an uncommon complication of COVID-198.
This case also serves as a warning for delaying care in acute myocardial infarction during this pandemic. There has been a significant decline in STEMI volume during COVID pandemic in US and Europe with an estimated 38% reduction in cardiac catheterization laboratory activation for STEMI9. Globally, there has been an alarming rise in the rare complications of untreated ACS such as ventricular septal defect from transmural infarcts, papillary muscle rupture, left ventricular aneurysm and thrombus formation, and acute heart failure; complications which were seen in the era predating advent of primary PCI10. Our patient waited almost a full day before presenting to the hospital for his myocardial infarction, possibly due to the fear of infection by going to the hospital during the pandemic. By the time he finally presented, much of the damage from the myocardial infarction was complete, however due to ongoing chest pain in a patient with cardiogenic shock, revascularization was attempted. The patient had already developed anterior Q waves on his ECG, the troponins were severely elevated, and the ejection fraction severely reduced. Perhaps the outcome would have been different if the patient presented earlier and received timely revascularization. Given these fears, the medical community must continue to advocate for patients to not delay care for symptoms of acute myocardial infarction. Additionally, the care of patients with COVID-19 is challenging due to potential exposure of all the medical staff members involved in the care with the virus. At our center, all personnel in the catheterization laboratory wore personal protective equipment including gown, gloves, eye shields, and N95 masks.
Conclusions
COVID-19 infection can complicate the management of acute coronary syndromes. The overlap in signs and symptoms between ACS and COVID-19 can make diagnosis and treatment difficult. In addition, COVID-19 infection may result in a prothrombotic state that contributes to further cardiovascular complications. Physicians should continue to educate patients to pay attention to the symptoms and seek timely treatment of acute myocardial infarction to reduce the morbidity associated with this pandemic.
Learning Objective
1. Clinical presentation of AMI along with COVID-19 illness can complicate diagnosis and management of the former.
2. Re-education of patients regarding acute chest pain and early presentation is crucial.
3. An acute infectious process can cause acute coronary syndrome (ACS) through acute inflammation, biomechanical stress and vasoconstriction. Much more is to be learnt regarding cardiovascular pathophysiology during COVID-19 illness.
References
- Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72314 cases from the chinese center for disease control and prevention. JAMA. 2020; DOI: 10.1001/jama.2020.2648. [epub ahead of print].
- Shi S, Qin M, Shen B, et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. 2020; 5(7): 802–810. doi:10.1001/jamacardio.2020.0950
- Kwong JC, Schwartz KL, Campitelli MA, et al. Acute myocardial infarction after laboratory-confirmed influenza infection. N Engl J Med. 2018; 378: 345-353.
- Corrales-Medina VF, Madjid M, Musher DM. Role of acute infection in triggering acute coronary syndromes. Lancet Infect Dis. 2010; 10: 83-92.
- Spiezia L, Boscolo A, Poletto F, et al. COVID-19-Related Severe Hypercoagulability in Patients Admitted to Intensive Care Unit for Acute Respiratory Failure. Thromb Haemost. Published online 4, 2020. DOI: 10.1055/s-0040-1710018
- Klok FA, Kruip MJHA, van der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thrombosis Research. 2020. Article in press.
- Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective study. The Lancet. 2020; 395 (10229): 1054-1062.
- Li, Yanan, Wang, et al. Acute Cerebrovascular Disease Following COVID-19: A Single Center, Retrospective, Observational Study (3/3/2020). Available at SSRN: https://ssrn.com/abstract=3550025 or http://dx.doi.org/10.2139/ssrn.3550025
- Garcia S, Albaghdadi MS, Meraj PM, et al. Reduction in ST-segment elevation cardiac catheterization laboratory activations in the United States during COVID-19 pandemic. J Am Coll Cardiol. 2020;Epub ahead of print.
- https://www.tctmd.com/news/dire-unusual-stemi-complications-blamed-covid-19-hospital-avoidance
